Scientists Discover the Hidden Cellular Code That Constructs Every Human Face
Summary
New research shows that human facial shape is influenced by cellular programs established very early in embryonic development. By combining detailed developmental studies in mice with large human genetic datasets, researchers linked specific genetic variation to distinct groups of cells that guide how the face forms. These findings help explain normal facial diversity and offer insight into congenital craniofacial conditions and genetic disorders that affect facial structure.
Who This Research Is Most Relevant For
This research is most relevant for conditions where early development and genetics play a central role in long-term structure, including:
Congenital craniofacial conditions such as cleft lip and cleft palate
Genetic syndromes that affect facial bones or connective tissue
Developmental facial asymmetry linked to early growth patterns
Autoimmune or inflammatory conditions that affect connective tissue or bone
Pediatric transplant and regenerative medicine contexts involving congenital abnormalities
Although this study focuses on facial development, its approach has broader relevance across developmental biology, autoimmune research, and transplant science. This research will be most meaningful for individuals and families navigating conditions tied to early development or tissue structure.
Key Findings at a Glance
Facial shape variation begins early in embryonic development
Groups of unspecialized cells carry region-specific positional programs
These programs guide how facial bones and connective tissue form
Human genetic variants linked to facial traits map to these same cell populations
The findings connect genetics, development, and congenital disease
What the Science Shows in Technical Terms
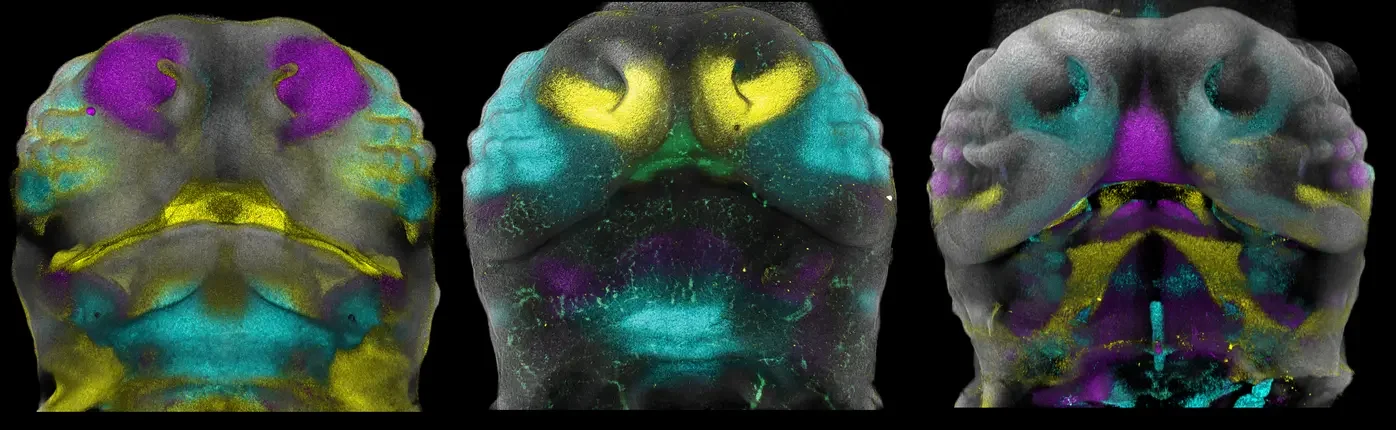
Researchers examined how facial tissue develops at the cellular level during early embryonic growth. They focused on mesenchymal cells, which are unspecialized cells that later form bone, cartilage, and connective tissue in the face.
Using single-cell gene expression analysis in mouse embryos, the team found that these cells are not uniform. Instead, they carry distinct positional programs, meaning each group of cells is biologically set up to form a specific region of the face. These programs are established before visible facial features appear.
The researchers then compared this developmental map with large human genetic studies that link DNA variation to facial traits. Many genetic variants associated with facial shape aligned with the same cell populations identified during early development.
Science Translated to Real-World Understanding
Researchers studied how the face forms very early in development by looking at individual cells that later become facial bones and tissue. They found that different groups of these cells already have instructions telling them which part of the face to build, long before facial features are visible. When they compared this with human DNA studies, the same genes linked to facial shape were connected to these early cell groups. This helps explain why small genetic differences early in development can have lasting effects on how tissues form and function over time.
Why This Matters for Your Health
This research helps clarify how genetic variation translates into physical structure. Many genetic variants associated with facial shape also appear in congenital craniofacial conditions, where development does not follow typical patterns.
By identifying the cellular pathways involved, these findings support more accurate interpretation of genetic test results and strengthen clinical understanding of why certain facial differences occur. This can improve diagnosis, counseling, and long-term care planning.
For patients and families, the research provides clearer biological explanations. For clinicians, it offers a more precise framework for connecting genes to anatomy. Understanding this connection can help people ask better questions, interpret genetic results more clearly, and feel more prepared when navigating care.
Autoimmune and Transplant Implications
This study does not directly examine autoimmune disease or transplantation, but its implications are relevant to both areas.
In autoimmune and inflammatory conditions, immune signaling can influence connective tissue, bone remodeling, and structural stability. Understanding how tissues are originally patterned during development helps explain why certain tissues may be more vulnerable to immune-mediated damage later in life.
In transplant and regenerative medicine, the findings highlight the importance of cell identity and developmental programming. Similar approaches are increasingly used to study tissue integration, long-term stability, and how tissues respond over time under immune suppression.
Where to Learn More
If this research raised questions about facial development, genetic conditions, or long-term tissue health, the following resources offer clear explanations, education, and support related to the topics discussed in this article:
Genetic and Rare Diseases Information Center (GARD)
A trusted, plain-language resource that helps individuals and families understand genetic conditions, testing results, and what research findings may mean for care.Cleft Palate Foundation
Provides education, family support, and care navigation resources for people affected by cleft lip, cleft palate, and other craniofacial differences.FACES: The National Craniofacial Association
Offers patient education, financial assistance guidance, and community support for individuals with craniofacial conditions.National Society of Genetic Counselors
Helps individuals find genetic counselors and understand how genetic information is used in medical decision-making.Children’s Craniofacial Association
Supports families navigating craniofacial diagnoses with education, peer connection, and care coordination resources.
These organizations help translate complex genetic and developmental science into practical understanding, emotional support, and informed care navigation.
Limitations to Keep in Mind
This research relies heavily on mouse developmental data to model human biology. While these models are highly informative, they do not capture every aspect of human development.
Facial structure is influenced by many genes and environmental factors, and no single study explains all variation. The findings describe biological mechanisms, not fixed outcomes.
What Researchers Are Studying Next
Future research will likely focus on:
Testing these developmental programs directly in human tissue
Expanding genetic datasets to include rarer variants
Studying how environmental and immune factors interact with early developmental pathways
Applying similar methods to other organs and tissues
Bottom Line
This study clarifies how genetic variation influences tissue structure by acting on specific cell populations early in development. For conditions involving craniofacial differences, connective tissue disorders, autoimmune disease, or transplanted organs, this framework strengthens how researchers and clinicians interpret genetic findings and tissue behavior over time. While not yet used in clinical care, this understanding supports more precise research and helps lay the groundwork for earlier, better-informed monitoring and care decisions over time.
Sources
Max Planck Institute for Evolutionary Biology, Transferred genetic programs shape facial development, 2025
Nature Communications, Murillo-Rincón et al., Positional programs in early facial development link genetic variation to human facial shape, 2025
FaceBase Consortium, National Institutes of Health
NIH-supported research initiative providing genetic and developmental data on craniofacial biology used broadly in this field.National Institute of Dental and Craniofacial Research, National Institutes of Health
Authoritative source for research on craniofacial development, genetic conditions, and facial structure across the lifespan.Annual Review of Genomics and Human Genetics, Genetic architecture of craniofacial morphology, 2022