Tegoprubart: A Next-Generation Anti-Rejection Therapy
Summary
A Phase 2 clinical trial studied a new anti rejection medication, tegoprubart, in people who had recently received a kidney transplant. After one year, kidney function was similar to that seen with tacrolimus, the most commonly used transplant drug today. Patients receiving tegoprubart experienced much lower rates of diabetes after transplant, neurologic side effects such as tremor, cardiovascular complications, delayed kidney function, and serious bacterial infections. Rejection occurred more often with tegoprubart, but kidney function remained preserved. The findings support further study rather than immediate changes to standard care.
Who This Research Is Most Relevant For
This research is most relevant for kidney transplant recipients concerned about long term medication side effects, particularly those who develop diabetes, tremor, or cardiovascular complications after transplant. It is also relevant for transplant clinicians evaluating alternatives to calcineurin inhibitor based regimens, as well as caregivers and families seeking to understand emerging transplant therapies.
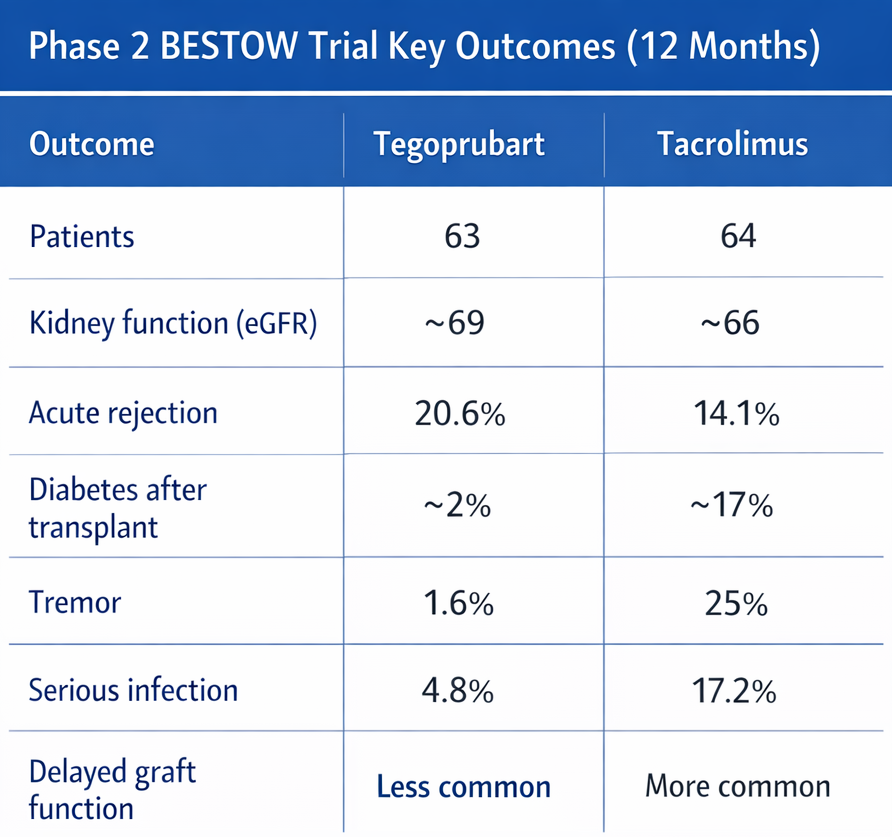
Key Findings at a Glance
Kidney function at 12 months was similar between tegoprubart and tacrolimus. New onset diabetes after transplant occurred far less often with tegoprubart, as did neurologic side effects such as tremor and serious bacterial infections. Rejection occurred more often with tegoprubart, but kidney function remained stable in those patients. Early recovery after transplant appeared smoother, with fewer days of dialysis when delayed graft function occurred.
What the Science Shows in Technical Terms
The BESTOW trial was a randomized, head to head Phase 2 study sponsored by Eledon Pharmaceuticals. It enrolled 127 adult kidney transplant recipients across 44 centers worldwide. Participants were randomly assigned to receive either tegoprubart or tacrolimus as the primary immunosuppressive agent. All patients received standard background therapy, including rabbit antithymocyte globulin induction, mycophenolate, and corticosteroids.
The primary endpoint was kidney function at 12 months, measured by estimated glomerular filtration rate, or eGFR. Secondary endpoints included biopsy proven acute rejection, delayed graft function, metabolic and neurologic adverse events, cardiovascular complications, and serious infections.
Science Translated to Real World Understanding
Both medications protected the transplanted kidney equally well over the first year. The meaningful difference was not kidney performance but how often patients experienced side effects that affect daily life and long term health. Although rejection occurred more often with tegoprubart, kidney function remained stable during the study period, suggesting these episodes were generally manageable.
Why This Matters for Your Health
Many transplant patients are told that side effects such as diabetes, tremor, or high blood pressure are unavoidable after transplant. This study suggests that some of these complications may be related to the medication rather than the transplant itself. Understanding this allows patients to engage more confidently in conversations about treatment options and long term care planning.
The Impact on the Future of Transplant Medicine
For people living with a transplant, this study offers real hope that protecting the kidney does not have to come at the expense of the rest of the body. By showing that kidney function can be maintained with fewer risks of diabetes, tremor, serious infections, and heart strain, and without the same degree of kidney toxicity associated with long-term tacrolimus use, this research points toward a future where life after transplant may be healthier, more stable, easier to sustain over time, and transplanted kidneys last longer.
Where to Learn More and Get Involved
Eledon Pharmaceuticals
The sponsor of the BESTOW trial. Their website posts official updates on tegoprubart, published trial data, and future clinical trial plans.
ClinicalTrials.gov
Search “tegoprubart” or “BESTOW kidney transplant” to view registered studies, eligibility criteria, participating transplant centers, and study contact information. This is the primary public gateway for trial involvement.
American Society of Nephrology Kidney Week (ASN)
ASN Kidney Week abstracts and session recordings provide expert discussion of new transplant data, including how clinicians are interpreting these findings in practice.
Your Transplant Center’s Research or Clinical Trials Office
Most transplant centers have a research coordinator. Asking whether your center participates in immunosuppression trials can help you understand current or upcoming study options.
UNOS and OPTN Patient Education Resources
These organizations provide guidance on patient rights, transplant research, and how clinical trials fit into transplant care decisions.
Limitations to Keep in Mind
This was a Phase 2 trial with a relatively small number of participants and one year of follow up. Long term kidney survival, chronic rejection outcomes, and effectiveness in higher risk transplant populations remain unknown. These findings should be interpreted as early but informative.
What Are Next Steps
The sponsor has indicated plans to advance tegoprubart into Phase 3 trials. These studies will include larger patient populations, longer follow up, and closer evaluation of rejection risk and long term kidney outcomes.
The estimated timeline if development continues successfully:
2026–2027: Phase 3 clinical trials begin and enroll larger transplant populations
2028–2030: Phase 3 trials complete with longer follow-up on kidney survival and rejection
2029–2031: FDA review and potential approval if results remain positive
Earlier access may occur before approval only through participation in clinical trials at select transplant centers.
Bottom Line
This study suggests that protecting a transplanted kidney may not require accepting the full burden of long term side effects many patients experience today. Tacrolimus remains the standard of care, but tegoprubart represents a credible step toward safer immunosuppression that warrants continued study.